Tag: semiconductor example
-
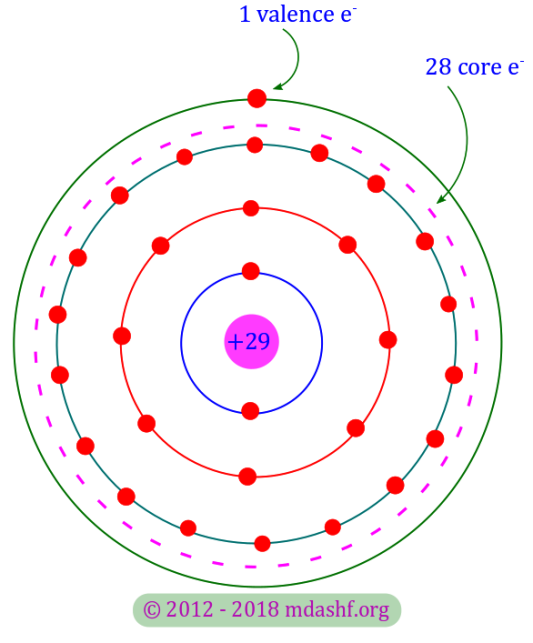
Semiconductors and charge carriers (L-I)
Analog electronics and applications Conductors, semiconductors and holes as charge carriers Topics covered in this lecture A. Conductors B. Semiconductors C. Holes D. Intrinsic semiconductors Conductors: A conductor is the name of a material which is a good conductor of electricity. Copper ( Cu ), Silver ( Ag ) and Gold ( Au ) are…