Analog systems and applications, lecture — I
“Conductors, semiconductors and holes as charge carriers”
This article belongs to a series of lectures on analog electronics, the paper goes by the name “Analog Systems and Applications” for the physics honors degree class. All lectures of this series will be found here. This is the first lecture of this series. This particular lecture was delivered on 2nd January 2018.

Topics covered in this lecture
A. Conductors
B. Semiconductors
C. Holes
D. Intrinsic semiconductors
Conductors
A conductor is the name of a material which is a good conductor of electricity. Copper ( Cu ), Silver ( Ag ) and Gold ( Au ) are examples of materials which are good conductors of electricity, in other words they are known as conductors.
A natural question arises as to why copper is a good conductor of electricity. Such a fact can be understood from its electronic configuration. Electronic configurations are a good way to understand the physical as well as chemical properties of materials.
A great deal of our modern understanding of materials and their properties are based on the detailed electronic configuration facts of the same.
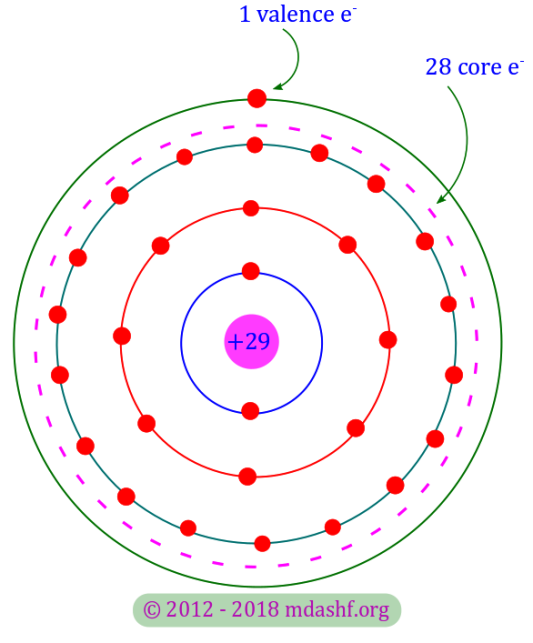
The copper has 29 electrons in its atom. That means it has an equal number of protons. It has two isotopes, one has 34 and the other has 36 neutrons. Isotopes are the same chemical element having 2 or more than 2 different types of nuclei, due to difference in the number of neutrons. As a whole copper atom is electrically neutral.
The 29 electrons are distributed into shells or orbits. Consequently the first orbit has 2, 2nd orbit has 8 and 3rd orbit has 18 electrons. There is only 1 electron in the outermost orbit of the copper atom.
The outermost orbit is known as “valence orbit“. The electrical properties of the atoms are determined by the valence orbit only.
The core of the copper atom consists of its nucleus — with total electric charge and inner orbits — with total electric charge
. Thus the core of the copper atom has a net electric charge of
and the valence electron has a charge of
.


Consequently the valence electron experiences a very small inward or attractive force from the core. Outside forces can easily dislodge the valence electron. The valence electron is thus called a free electron. Due to this reason copper behaves as a very good conductor of electricity.
Semiconductors
The best conductors such as copper, silver and gold have only one valence electron. By contrast the best insulators have 8 valence electrons.
Germanium ( Ge ) and Silicon ( Si ) are examples of semiconductors each with 4 valence electrons. Let us study the silicon electronic configuration.
A single silicon atom has 14 protons and 14 electrons.
The first orbit of silicon has 2 electrons. The second silicon orbit has 8 electrons. Thus the valence orbit of silicon has 4 electrons. Consequently the core of the silicon has a net charge of , as the silicon has 14 protons and 10 electrons in the inner orbit.


The 4 valence electrons make silicon a semiconductor. We will understand the deeper significance of this when we study the band gap concept in various kind of materials, viz. metals, semiconductors and electrons in our solid state physics lecture, here, as well as some later lectures of this series, i.e. analog electronics lectures, all of which are to be found here.
In a solid each silicon atom shares its electrons with 4 neighboring atoms. Thus each silicon atom in a silicon crystal lattice, has 8 valence electrons, due to sharing. It depends upon the lattice properties of silicon as a crystal in it solid state configuration.
The valence electron no longer belongs to any single atom, its now shared among many neighboring atoms. Due to interaction between two neighboring cores in the silicon crystal structure, each shared electron experiences an equal and opposite force. This gives rise to a covalent bond which is the reason why the solid crystal is held together.
There is always a tendency in the outer orbit of all elements to have 8 valence electrons. This is known as “valence saturation” If there is a deficiency of valence electrons, elements combine and share electrons so that there are 8 valence electrons for every atom. Valence saturation is what the atoms prefer for their stability.
A silicon crystal is almost a perfect insulator at room temperature — , as there are 8 valence electrons.
Holes
Due to thermal excitation the electrons in the silicon atoms might gain some energy, which is sufficient to dislodge it from the valence orbit. The electron goes to a larger orbit where it is essentially free, a condition for increase in electrical conductivity.
As a result there is a deficiency of 1 electron in the valence orbit. This deficiency is given the name: hole.
At room temperature there are only a few number of electrons and holes, i.e. the number of dislodged electrons and consequent creation of holes is small in number. For a pure semiconductor the number of electrons — that are now free and holes, produced due to thermal excitation is equal. When a free electron approaches a hole they can recombine — to create an electron, that is no more free as it was previously.
Intrinsic semiconductors
An intrinsic semiconductor is a pure semiconductor. Every atom in the intrinsic semiconductor is of the same type. Under the influence of external electric force, e.g. an applied voltage, electrons and holes drift in opposite direction.

The free electrons and holes are known as “charge carriers” or simply carriers — or free carriers, as they carry charge from one place to another. The electrons and holes flow in opposite directions. The net flow of charge in the semiconductor is the combined effect of “flow of free electrons” and “flow of holes“.

a. In an electric circuit, shown above, electrons will drift to left and go out of the semiconductor to the positive potential.
b. Holes will move to the right but can’t come out of the semiconductor.
c. Holes will recombine with electrons coming from the negative potential and entering the semiconductor, on the right.
Leave a comment