Mindblown: a blog about philosophy.
-
The Bohr model of the Hydrogen atom: atomic spectra and quantization, Quantum Lecture – 4 and 5.
Light is known to have possessed various colors. It would be more appropriate to state that light and color are two entirely distinct entities. Light does not contain color just like you do not contain your spouse. Its another matter you both appear at the same venue on the basis of the same invitation card.…
-

I ain’t no meek heart
I ain’t no meek heart >> And I won’t date the vile Would you date the vile? Compile cos once in a while you gotta go the extra mile so smile don’t change your style everyone will be friends with you if you are ductile all of’em hate the rigid the hot headed pedophile they…
-
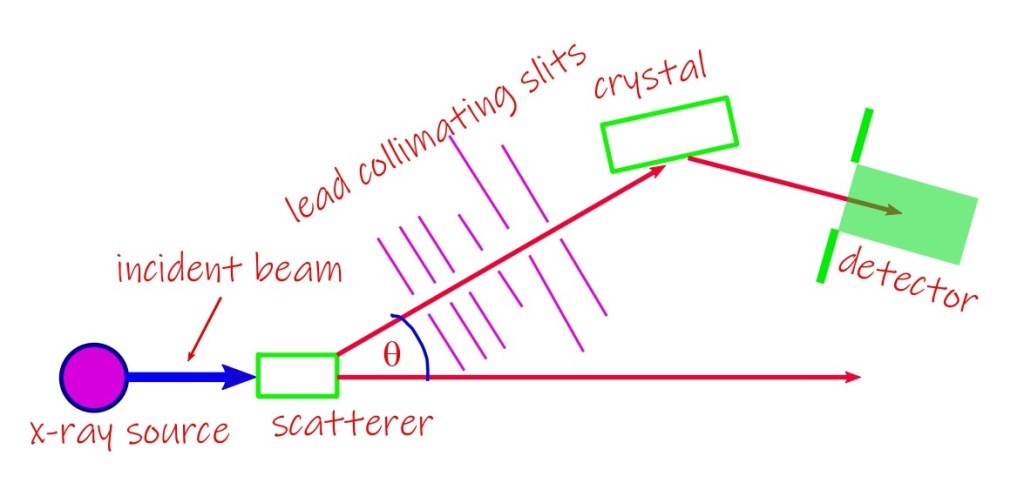
Compton effect, Quantum Lecture 3.
As we have mentioned earlier, we would be discussing a few experiments that elucidates the nature of the new theory based on the ideas of quantization and wave-particle duality. This lecture will be an addition to the phenomenon of photoelectric effect that we discussed in our last lecture, as an example of how a classical…
-
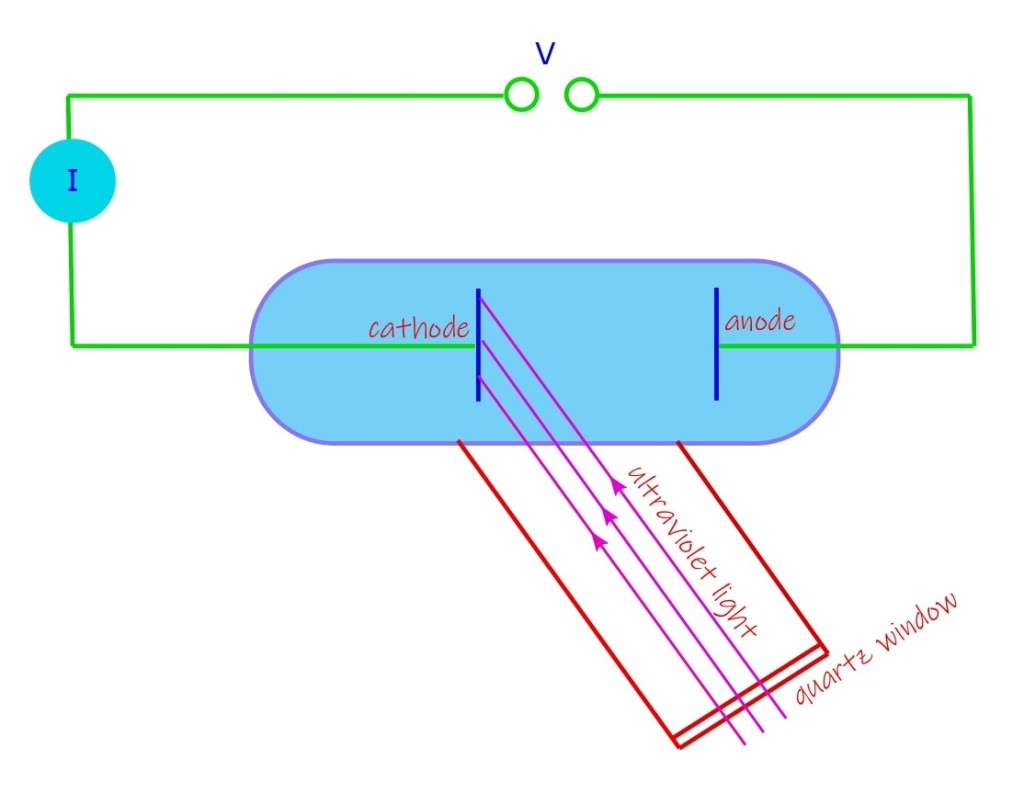
Photoelectric Effect, Quantum Lecture 2.
In 1886-89, (at the age 29-31, and deceased barely at the age 36) Heinrich Rudolf Hertz, a German physicist, performed experiments in which he conclusively established the existence of electromagnetic waves, proposed by the theoretical works of James Clerk Maxwell, a Scottish scientist who unfortunately did not live to see this. Maxwell lived only for…
-

Black body radiation, Quantum Lecture 1.
This article belongs to a group of lectures I intend to prepare for their online dissemination — no hand written notes of the same is available as I did not deliver these lectures, its an attempt at on-the-fly preparation of the concepts, exclusively available at this website. This series is on quantum mechanics and bears…
Got any book recommendations?